- Establish a strategy that meets regulatory requirements from agencies like the FDA and EMA, and other regulatory agencies.
- Regulatory submission: Compiling the necessary data on the drug’s quality, manufacturing process, stability, and analytical methods.
- cGMP clinical and commercial manufacturing and process design: Optimizing and validating the processes for producing the drug product, from early-stage development to commercial scale-up.
- Quality assurance and compliance: Ensuring that all practices meet Good Manufacturing Practice (GMP) and other compliance standards.
- Conduct cGMP/GLP/CGP audits in compliance with FDA and EMA and other agencies.
- Process optimization in Upstream and Downstream, DoE design, Process Performance Qualification, and Risk Assessment.
- Clinical and commercial cGMP manufacturing, manufacturing batch records and trouble shooting.
- cGMP facility design with flexible and modular in compliance with US FDA and EU GMP standards.
Overview services:
CMC in Drug Development and Life Cycle Management
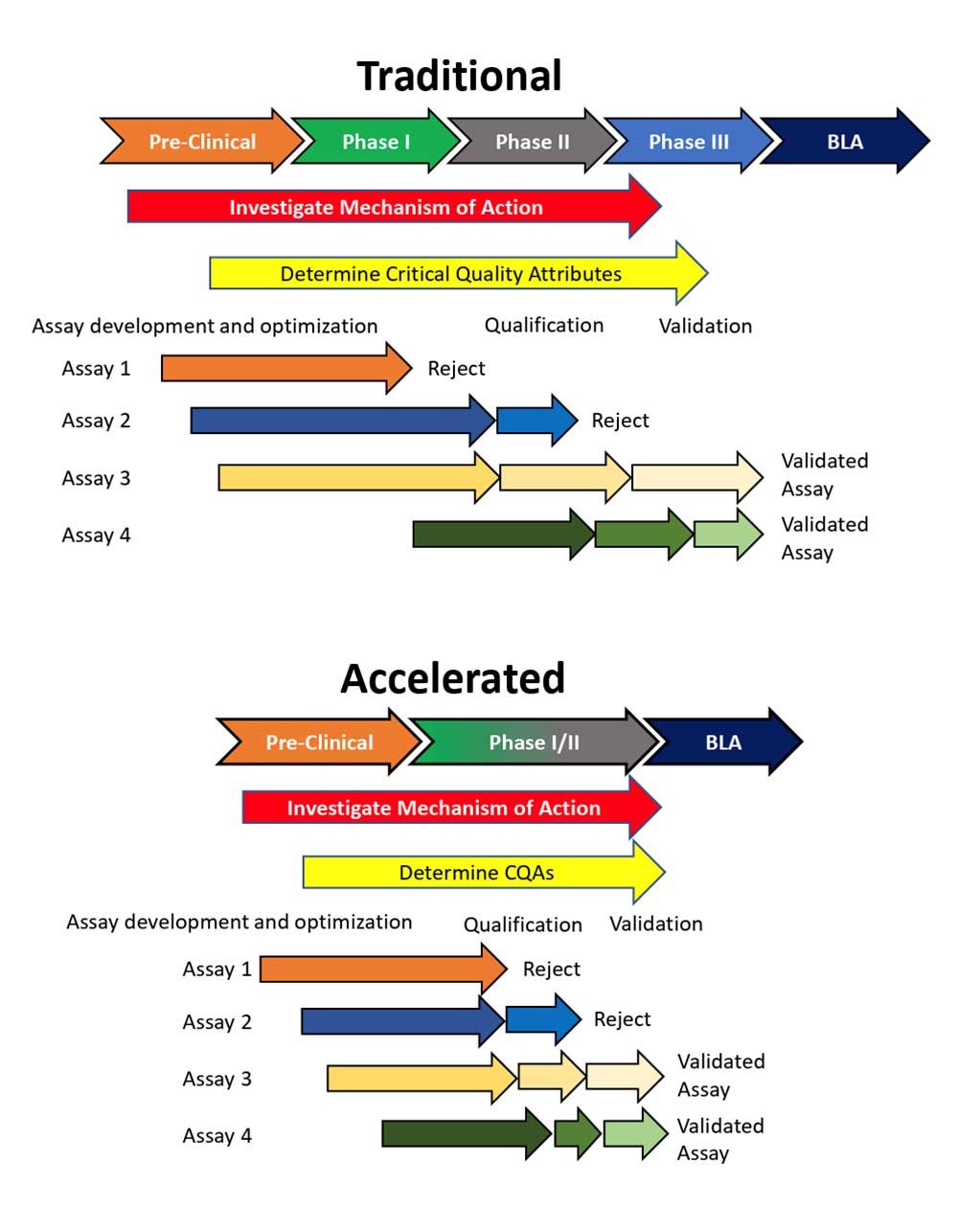
Accelerated Clinical Development
Promising clinical results and accelerated clinical studies allow less time for product development
CMC: IND-enabling
- Pharmaceutical Quality
- CMC Requirements for INDs
- Drug Substance
- Drug Product
- Impurities
- Specifications
- Stability
- CMC Safety Concerns
The Pre-BLA Phase
- Clinical Data
- CMC Information
- Nonclinical Data
- Labeling
- Manufacturing changes and comparability
- Process validation, Assay validation
- Potency assays
- Readiness of manufacturing facilities
- Extractables and Leachables
NDA/BLA Submission
The BLA itself is typically organized into different modules, following the structure of the Common Technical Document (CTD)
- Module 1: Administrative Information and Prescribing Information
- Module 2: Summaries (including the Quality Overall Summary, Nonclinical Overview, and Clinical Overview)
- Module 3: Quality (CMC information)
- Module 4: Nonclinical Study Reports
- Module 5: Clinical Study Reports
Request for service
Contact us by filling form below or send us at info@vertexbiopharm.com
