- Asset scouting: Identifying and evaluating potential in-licensing opportunities and partners.
- Strategic advice: Determining which products to license and developing a comprehensive strategy.
- Deal structure and valuation: Negotiating upfront payments, milestones, and royalties to ensure fair value.
- Due diligence: Conducting thorough technical, financial, and IP reviews of the potential target.
- Negotiation: Leading discussions and finalizing the term sheet and full licensing agreement.
In-licensing and out-licensing structure
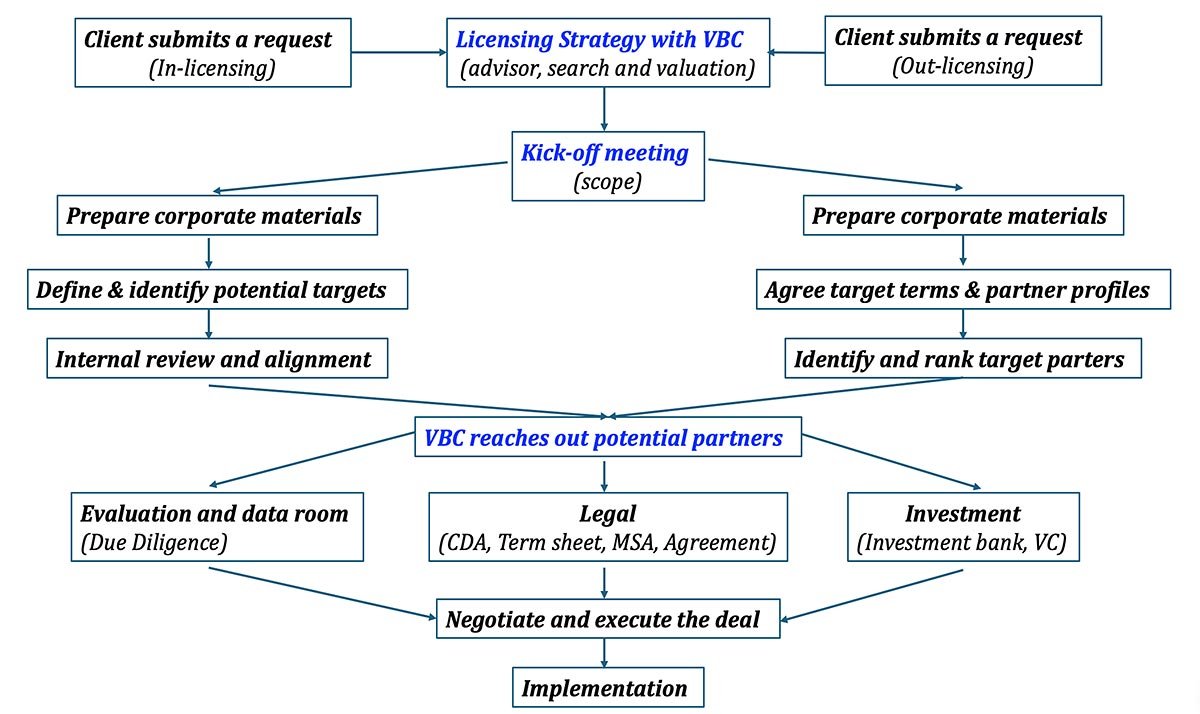
In-licensing services
Out-licensing services
- Partnership identification: Scouting for companies (often larger pharmaceutical firms) with the right therapeutic focus and capabilities.
- Pitch deck development: Creating compelling presentations to attract potential licensees.
- Strategic guidance: Advising on the best way to structure the deal and determine optimal licensing terms.
- Negotiation support: Helping to negotiate terms and conditions for the licensing agreement.
- Post-deal support: Assisting with the implementation of deal terms after the agreement is signed.
Current platform technologies available for out-licensing
- Three platforms of LNP/nanobody display (for mRNA, circular RNAs, DNA, peptide) for in vivo CAR-T, cancer vaccine, vaccine and others, with global rights
- AVV single plasmid platform with global rights
- AAV tissue-specific delivery with global rights
Current clinical assets available for out-licensing
- Clinical asset Phase I trial in solid tumors (small molecule, global rights)
- Clinical asset Phase II trial in solid tumors (small molecule, global rights)
- Clinical asset Phase III trial in solid tumors (small molecule, global rights)
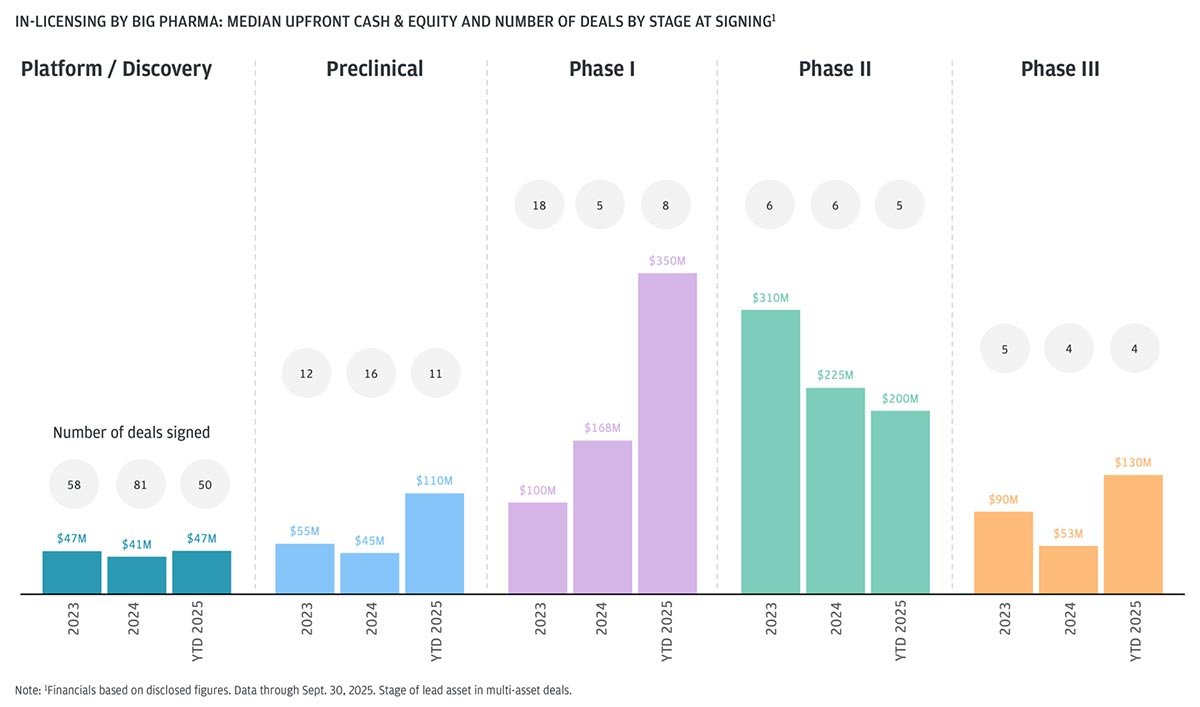
Big pharma made larger deal moves in Phase I as competition for key programs increased
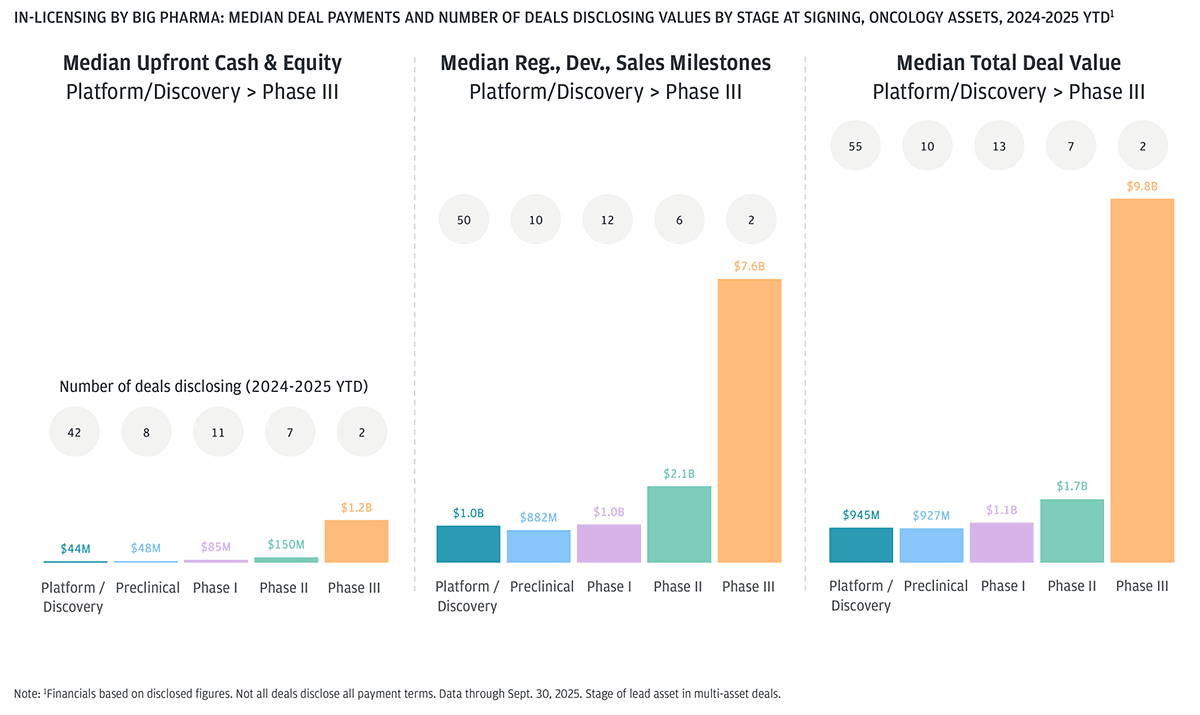
Oncology deal terms: Big pharma paying more in upfronts and milestones for later-stage assets
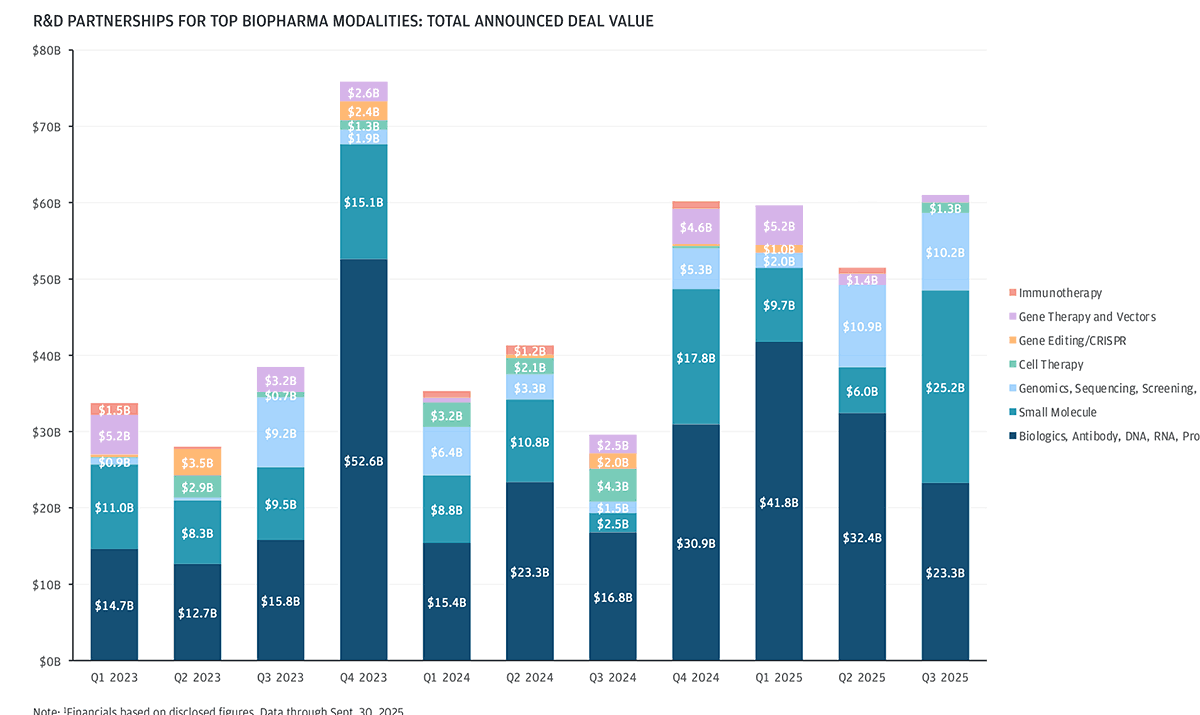
Biologics and small molecules have remained the top modalities for R&D partnerships
AVAILABLE OUT-LICENSING TECHNOLOGIES
Three platforms of LNP/nanobody display
Three platforms of LNP/nanobody display (LNP#1, LNP#2, LNP#3 for mRNA, circular RNAs, DNA, peptide) for in vivo CAR-T, cancer vaccine, vaccine and others, with global rights
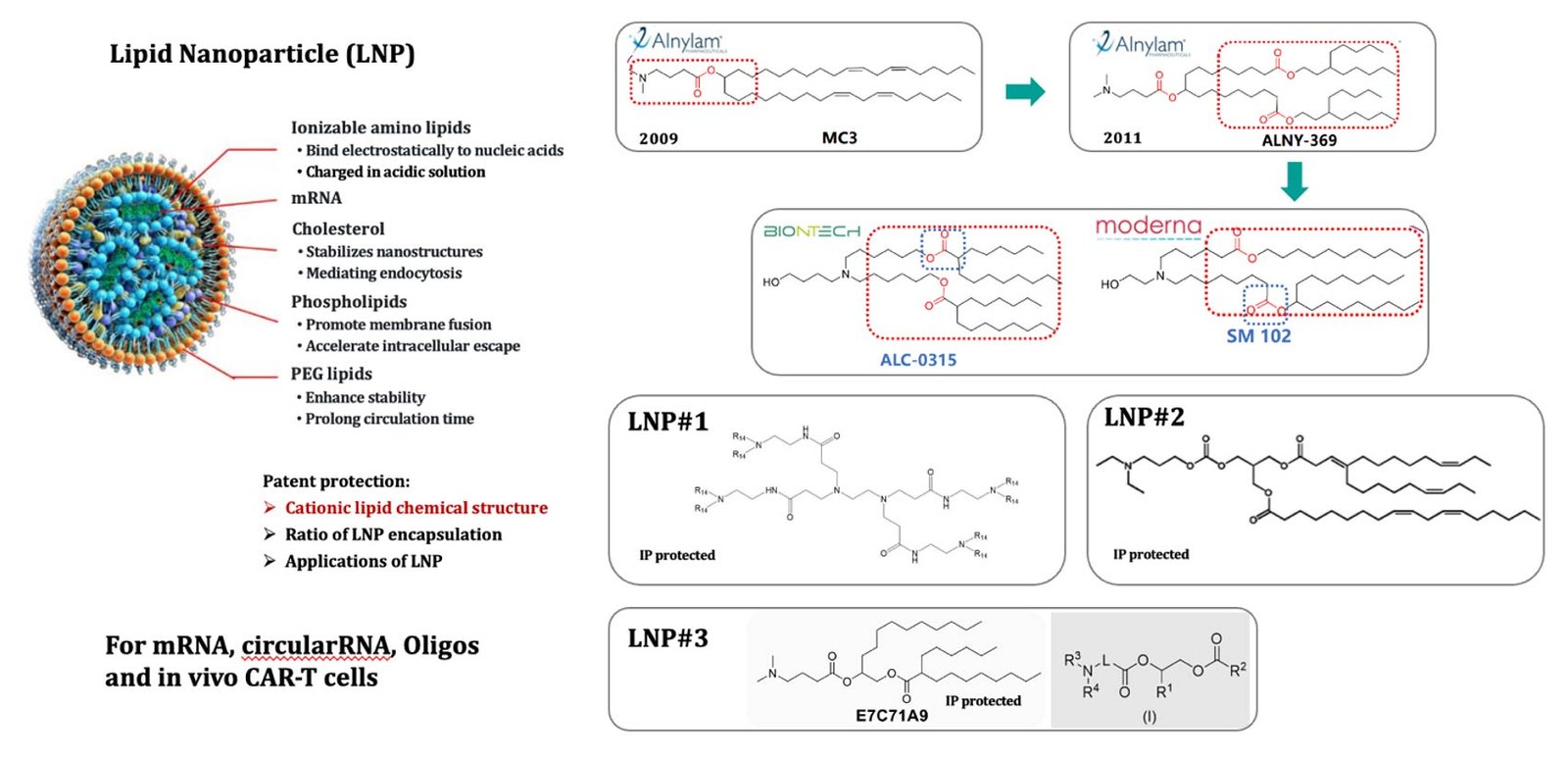
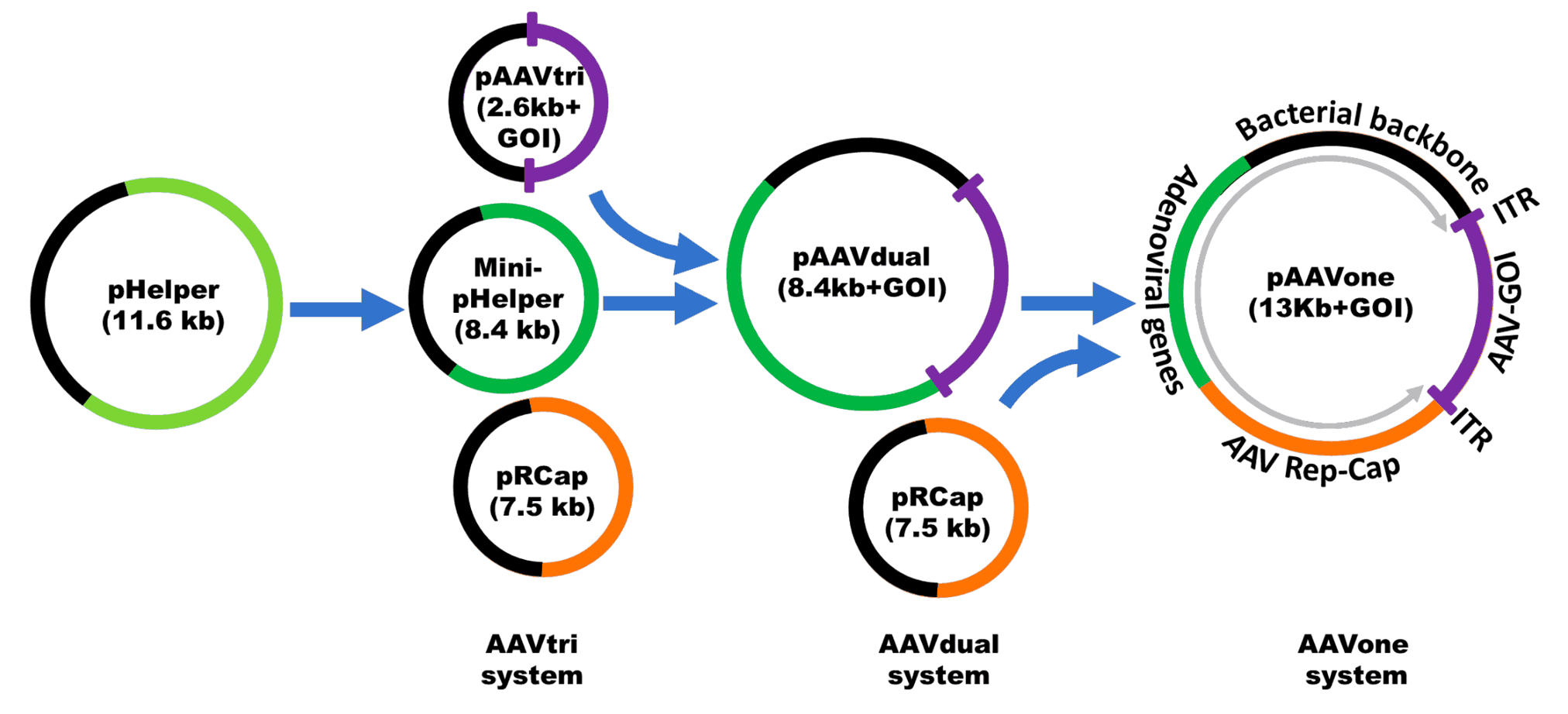
AAV single plasmid platform technology can be used for pipeline development with global rights
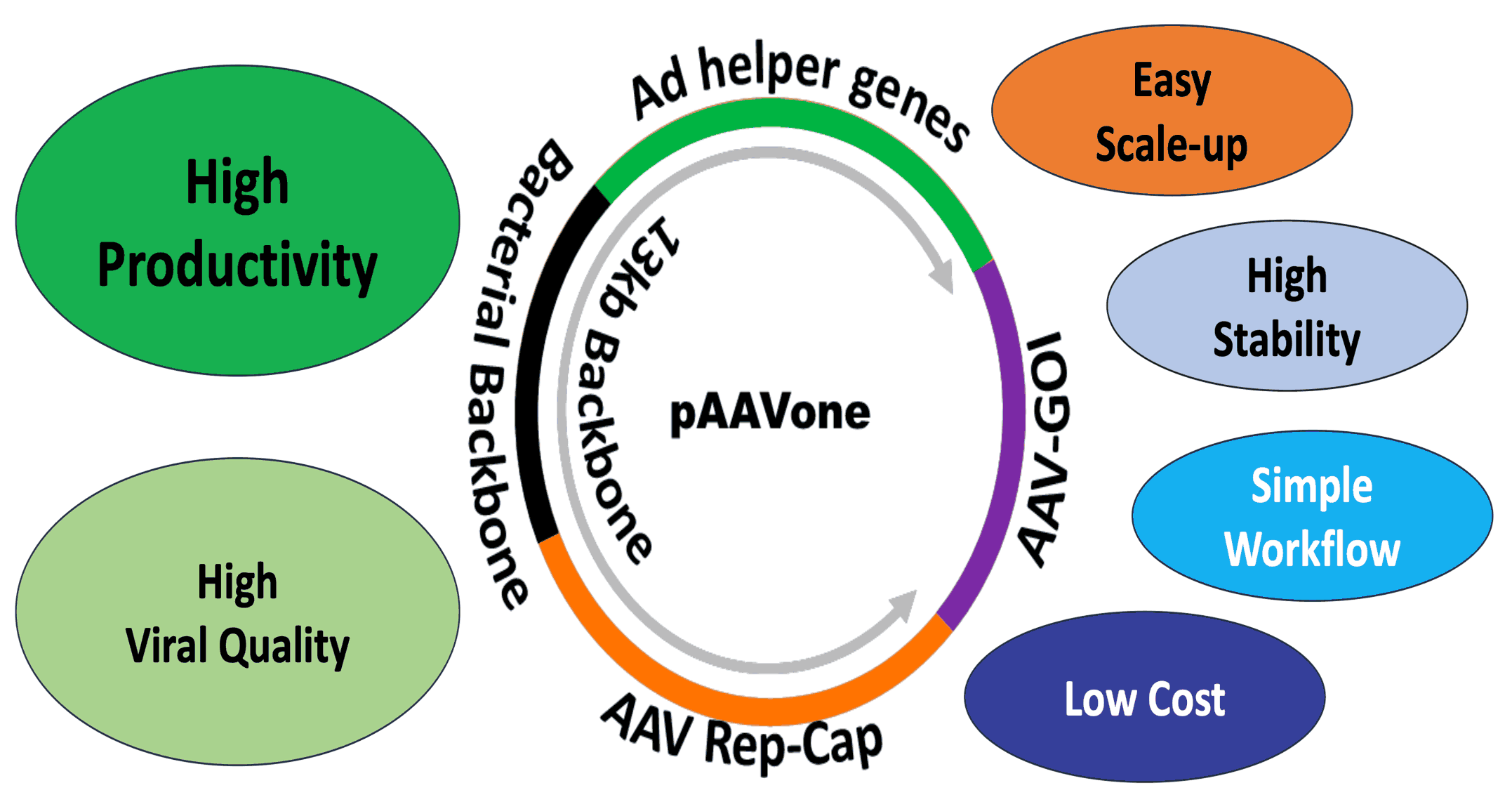
AAVone®: Streamlined Single-Plasmid AAV Production System
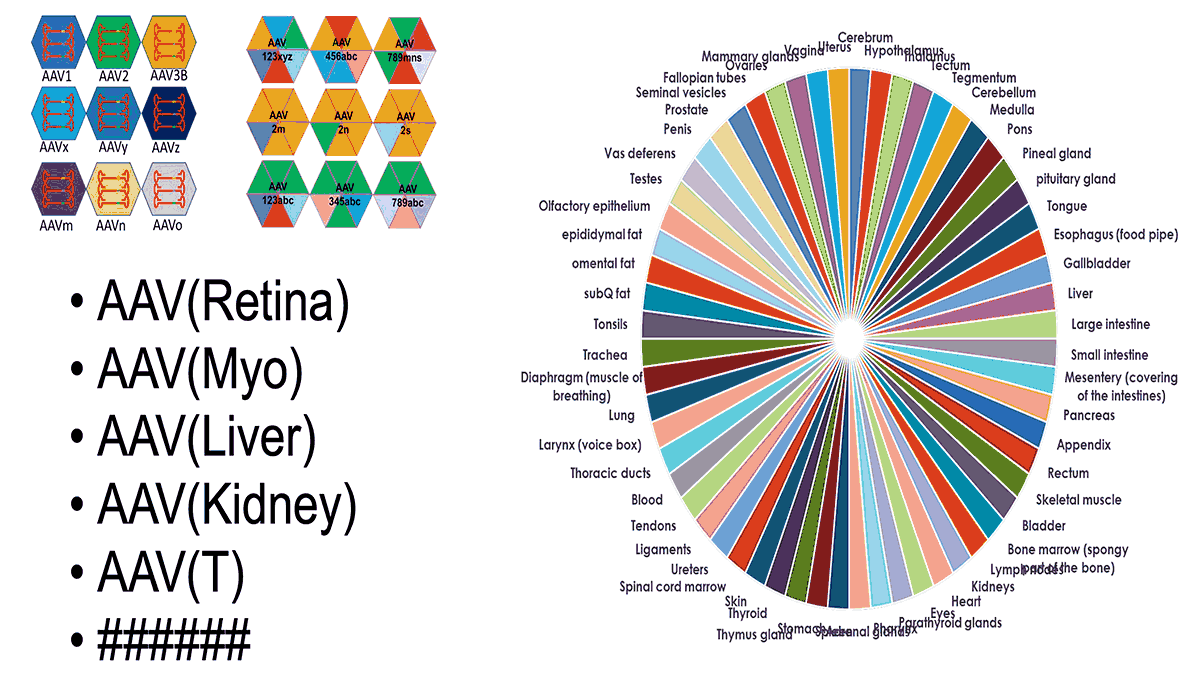
AAV tissue-specific delivery with global rights
Contact us for more information about tissue that you are interested in using AAV for the delivery. Currently, we have established AAV targeted for brain, lung, spinal cord, retina.
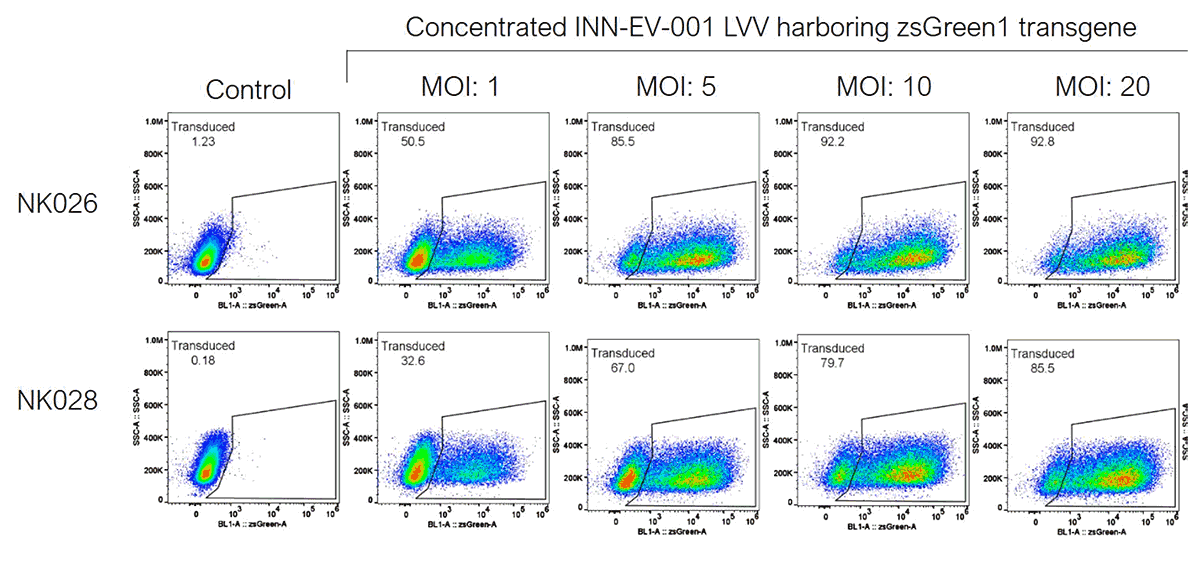
MPMV-Rless pseudotyped Lentiviral Vectors for In Vivo Gene Delivery, efficiency delivery for NK and T cells
Request for in/out licensing
Contact us by filling form below or send us at info@vertexbiopharm.com
Asset Identification:
- Name and description of the clinical asset (e.g., drug candidate, biologic, medical device).
- Therapeutic area and mechanism of action (MOA).
- Current development stage (e.g., pre-clinical, Phase 1, Phase 2, registration, or already marketed).
- Relevant intellectual property status, including patents and copyrights.
- Regulatory status (e.g., existing IND application, previous communications with the FDA or other regulatory bodies).
Clinical and Scientific Data:
- Summary of all conducted pre-clinical and clinical studies, including protocols, results, and safety data.
- Information on the unmet medical need the asset addresses and its differentiation from existing standard-of-care treatments.
- Documentation of clinical proof-of-concept (POC), including Phase 2 data if available, which is a key value inflection point.
Commercial and Market Potential:
- Target patient population and estimated market size.
- Competitive landscape analysis (efficacy, safety, dosing, cost).
- Proposed target market(s) or geographic territory for the license.
Logistics and Manufacturing:
- Manufacturing considerations (e.g., special handling, controlled substances).
- Stability data on the medicinal product and supply chain details.
- Proposed Deal Terms:
- Requested financial structure (e.g., upfront payment, milestone payments, royalties).
- Proposed license duration and other key terms and conditions.
