- Goal
- Maximize Productivity: Achieve high product titers (concentration).
- Ensure Quality: Control critical quality attributes (CQAs) of the therapeutic product.
- Enable Scale-Up: Develop a robust, reproducible process for manufacturing.
- Support Regulatory Submissions: Generate data for regulatory agencies (e.g., FDA).
VBC provides advisory consultation in process design and trouble shooting
- Cell Line & Vector Development: Selecting and engineering the most productive host cells and expression systems.
- Media & Feed Optimization: Designing nutrient-rich media and feeding strategies to maximize cell growth and product output.
- Bioreactor Process Development: Optimizing cultivation conditions (temperature, pH, aeration, agitation) in bioreactors.
- Scale-Up: Translating lab-scale processes to larger manufacturing scales.
- Process Understanding: Characterizing nutrient consumption, byproduct generation, and quality attributes.
- CDMO: Suggestion for right CDMO partners.
Process Development, Process Performance Qualification and cGMP Manufacturing
Upstream (USP) Support for Drug Substance (DS)
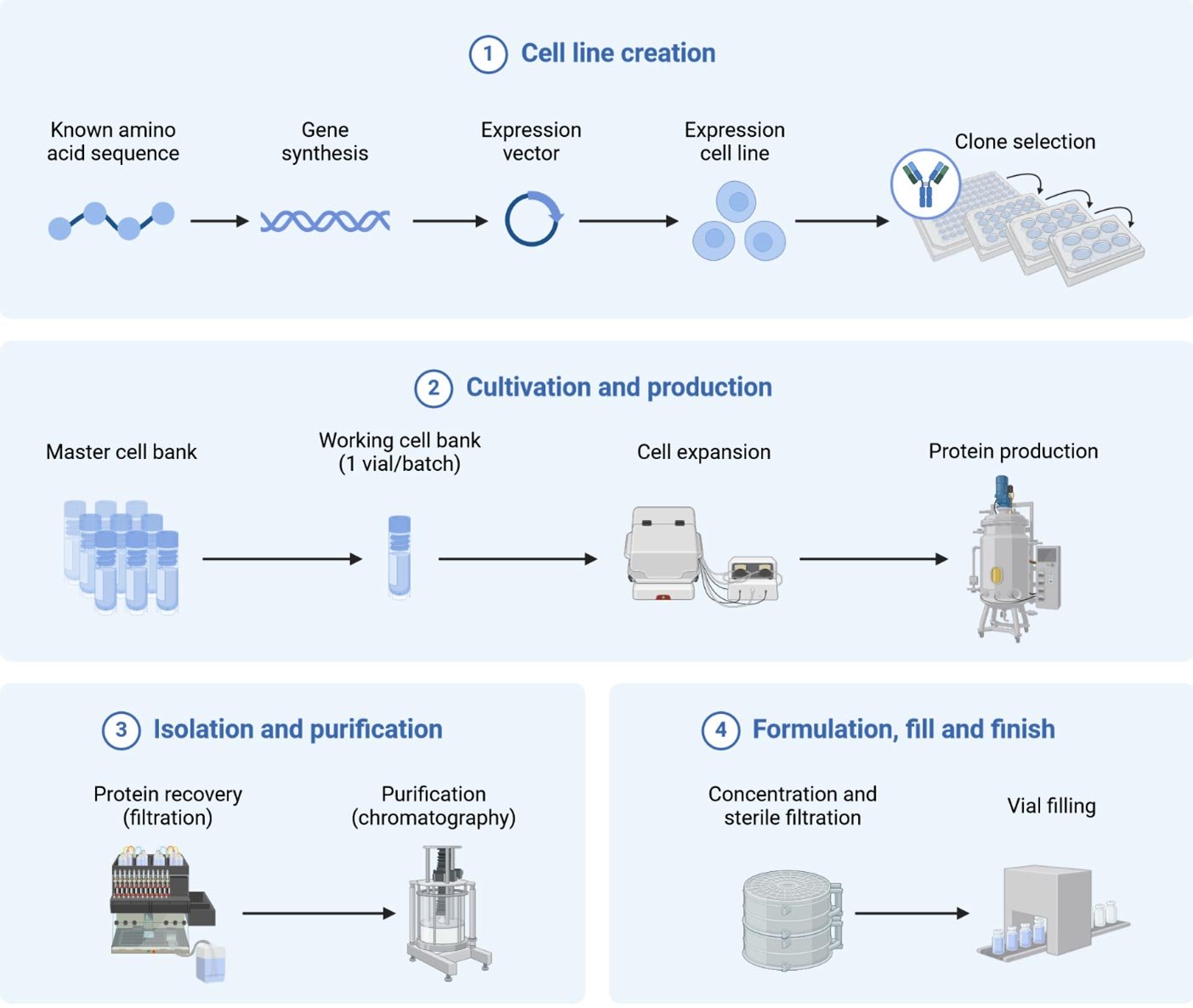
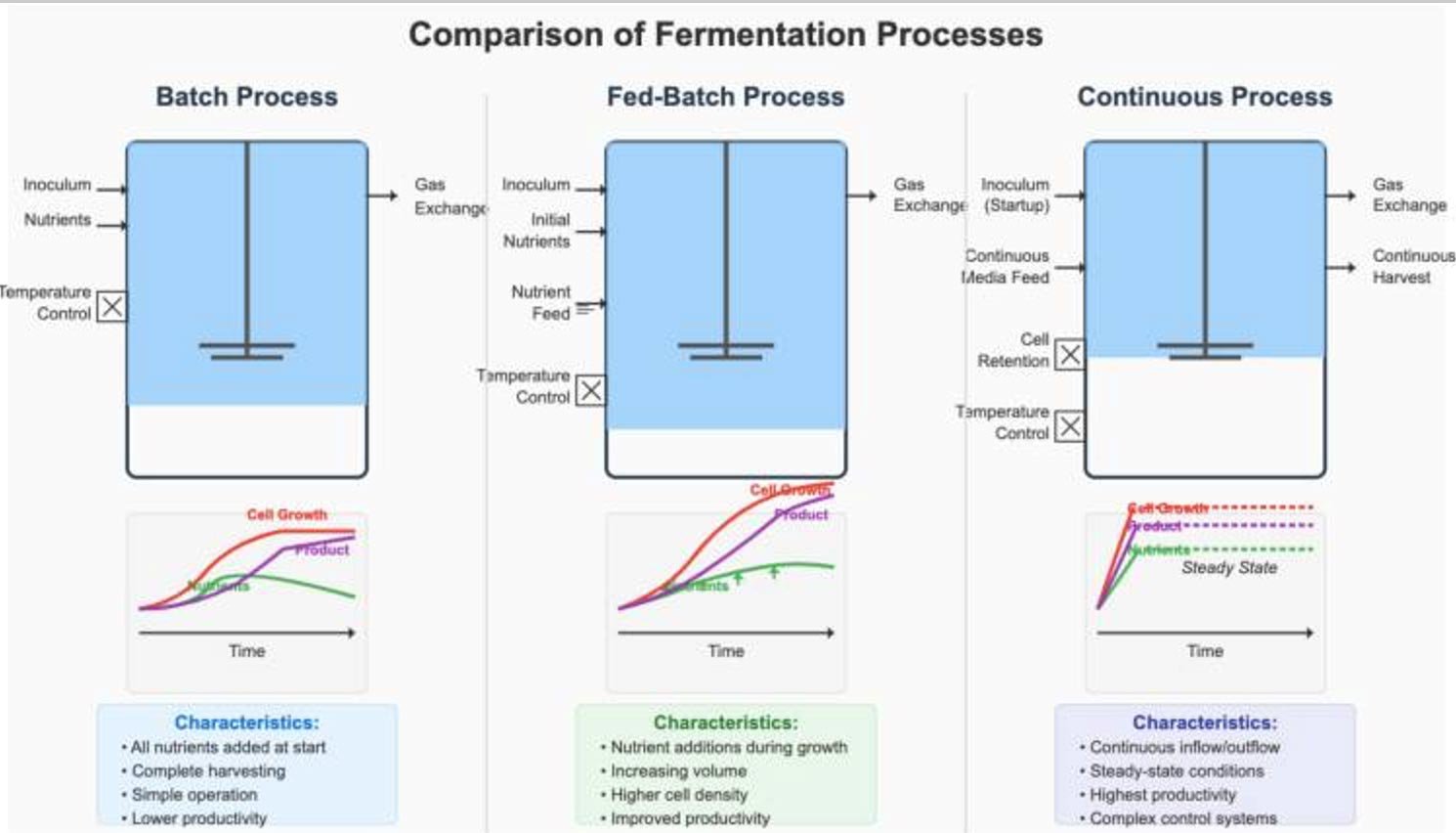
Downstream (DSP) Support for Drug Substance (DS)
- Goal
- Bottleneck Resolution: DSP is often a major bottleneck in bioprocessing; efficient development is crucial for industrial viability.
- Cost & Quality: It significantly impacts the final product’s cost, purity, safety, and efficacy, especially for high-value biologics like monoclonal antibodies, vaccines, and gene therapies.
- Scalability: Processes must be developed to scale seamlessly from lab bench to commercial manufacturing.
VBC provides advisory consultation in process design and trouble shooting
- Cell Separation/Harvest: Separating cells (biomass) from the liquid culture medium using centrifugation or filtration.
- Cell Disruption (if needed): Breaking open cells for intracellular products (e.g., homogenization, sonication).
- Clarification: Removing insoluble debris and impurities (e.g., depth filtration, centrifugation).
- Isolation & Purification: Using techniques like chromatography (affinity, ion exchange, size exclusion) and ultrafiltration/diafiltration (UF/DF) to separate the target molecule from similar impurities.
- Concentration & Polishing: Further concentrating the product and performing final purification steps (e.g., viral filtration, sterile filtration) to meet quality standards.
- Final Formulation & Drying: Preparing the product for stability and delivery (e.g., lyophilization, spray drying).
- CDMO: Suggestion for right CDMO partners.
Analytical and Assay Development: VBC Provides Technical Design and Analysis
Analytical development assays are crucial, highly specific methods created to identify, quantify, and characterize drug substances and products, ensuring their identity, purity, potency, and stability throughout the drug lifecycle, from discovery to manufacturing, following strict regulatory (ICH, FDA) guidelines like accuracy, precision, specificity, and robustness.Application in Drug Development: Used for quality control (QC), stability studies, ensuring product release, and supporting regulatory submissions (CMC sections).Types of modalities: biologics, including mAbs, proteins, cell and gene therapy products, and vaccines, as well as small-molecule drugs
Key Performance Characteristics (ICH Guidelines):
- Accuracy: How close results are to the true value.
- Precision: Repeatability and consistency of results.
- Specificity: Ability to measure the target without interference.
- Linearity & Range: Measuring across a concentration spectrum.
- LOD/LOQ: Limits of Detection and Quantification.
- Robustness/Ruggedness: Performance under slight variations (e.g., different analysts, instruments).
Types of assays that VBC provides advisory consultation in process design and trouble shooting
- Chromatographic: HPLC for separation and quantification
- Bioassays: Cell-based, enzyme activity assays for functional potency.
- Immunoassays: For biologics and specific targets.
- Viral titer assays: qPCR, ddPCR, TCID-50
- Purity assays: SDS- PAGE, Capillary Electrophoresis, HPLC, LC-MS/MS
Fill-Finish Support for Drug Product (DP)
- Goal
- Patient Safety: Prevents microbial contamination, which can cause harm or recalls.
- Product Efficacy & Stability: Ensures the drug remains potent and stable until use.
- Regulatory Compliance: Meets stringent Good Manufacturing Practices (GMP) standards.
- Complexity: Increases with new biologics, personalized medicines, and advanced delivery systems (e.g., pre-filled syringes).
VBC provides advisory consultation in process design and trouble shooting
- Preparation & Tech Transfer: Gathering product info (stability, pH, formulation) and tech transfer documents from early development.
- Engineering Runs: Testing the process on automated lines (small-volume or high-speed) in controlled environments (isolators) to validate equipment and protocols before GMP runs.
- Formulation: Preparing the bulk drug product (e.g., mixing, thawing, dissolving) in cleanrooms, with precise measurements and mixing times.
- Aseptic Filling: Transferring the sterile drug into containers (vials, syringes) using sanitized equipment, often with peristaltic or piston pumps, under strict sterility.
-
- Sealing: Securely stoppering, capping, and sealing containers to maintain sterility and integrity.
- Inspection: Performing 100% visual inspection for defects, particulate matter, and proper sealing.
- Labeling & Packaging: Applying labels and packing into secondary/tertiary packaging for distribution.
- Quality & Release: In-process testing, final release testing (microbiology, chemistry), quality assurance review, and batch release for shipment.
- Labeling: Applying clear, accurate information, including serial numbers for track-and-trace.
- Unique Identification: Creating and printing a unique serial number, lot, batch, and expiration date on primary (bottle/blister), secondary (carton), and tertiary (pallet) packaging.
- Secondary Packaging: Placing primary packages into cartons, often with patient information leaflets (PILs).
- Track & Trace: Linking these identifiers through aggregation, so a bottle’s code links to its carton’s code, and so on, allowing full supply chain visibility.
- Line Trials & Validation: Testing processes on the production line to ensure speed and quality before full-scale manufacturing.
- Distribution & Supply Chain: Arranging storage and shipping, with ongoing monitoring (Phase IV).
- CDMO/CPO: Suggestion for right CDMO partners.
